A new review in The Lancet Neurology, co-authored by Rosa Rademakers and an international team of experts, provides a comprehensive overview of amyotrophic lateral sclerosis (ALS) caused by C9orf72 hexanucleotide repeat expansions. The paper highlights major advances in clinical and molecular research, discusses the challenges of developing effective therapeutics, and outlines future research priorities.
Co-led by Adrian Isaacs of the UKDRI and Rosa Rademakers, the new review is part of a series of papers on genetic ALS, offering a broad perspective on the molecular underpinnings and potential treatment strategies for different genetic forms of the disease.
Complexity and convergence
Since the discovery of the C9orf72 repeat expansion 14 years ago, researchers have made significant progress in understanding its clinical and molecular effects. The mutation leads to both loss-of-function (reduced C9orf72 protein) and gain-of-function (toxic RNA and protein accumulation) mechanisms, affecting neurons and glial cells in the central nervous system and beyond. Despite this complexity, TDP-43 proteinopathy remains a key pathological hallmark in most affected individuals.
One of the major challenges in studying C9orf72 ALS is the variability in disease onset and progression. While some individuals carrying the mutation develop ALS or frontotemporal dementia (FTD), others remain asymptomatic for much of their lives. Understanding the genetic and environmental factors that influence this variability is a major area of research.
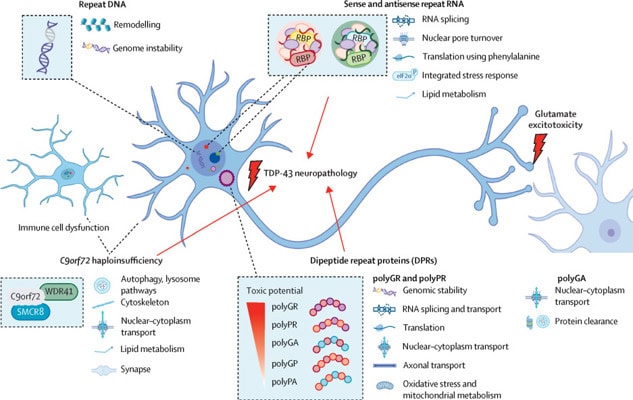
Accelerating therapeutic development
The first wave of therapeutics, particularly antisense oligonucleotides (ASOs) targeting sense C9orf72 repeat RNA, have not been successful in stopping neurodegeneration. This underscores the need to identify additional pathological drivers and develop more comprehensive therapeutic strategies. Future treatments must consider:
- Targeting both sense and antisense repeat-derived RNAs and proteins
- Exploring downstream effects of C9orf72 mutations in neurons and immune cells
- Developing biomarkers to track disease progression and treatment response
In the review Rademakers and her co-authors outline key research priorities to accelerate the development of effective therapies:
- Understanding genetic and environmental factors that affect disease onset and penetrance
- Developing TDP-43 neuroimaging and biofluid biomarkers to track disease progression
- Implementing clinical trial platforms that allow for testing multiple therapies simultaneously, improving efficiency and comparability
